 One can use the periodic table to predict whether an. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. Use the periodic table to determine the charge in an element First. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. Non-metals tend to gain electrons from metals. F gains 1 e - and Ogains 2 e -'s, and O gain e -'s to attain theconfiguration of Ne thus, the halogens tend to -1 charges and theoxygen family tends to a -2 charge. Metals tend to lose electrons to non-metals. On the other side of the periodic table elements gain electrons toresemble the next higher noble gas. Thus, the group 2 metals tend tohave a +2 charge. The alkali earth metals (group 2), such as, Mg or Sr lose two e-'sto attain the configuration of Ne. Valence is the number of connections an atom tends to form. Since K loses one electron (1 negative charge)it is no longer neutral it now has a +1 charge. The relationship between valence electrons and charge of an ion is EXTREMELY IMPORTANT. 4H 4 H atoms 4 × 1 4 4 × 1 4 valence electrons. In the case of the ammonium ion: 1 N 1 N atom 5 5 valence electrons. Kloses an electron so that it will have the same electronconfiguration as Ne. When drawing the Lewis structure of a polyatomic ion, the charge of the ion is reflected in the number of total valence electrons in the structure. The alkali metals will lose anelectron to resemble the next lowest noble gas thus, all the alkalimetals form +1 ions. 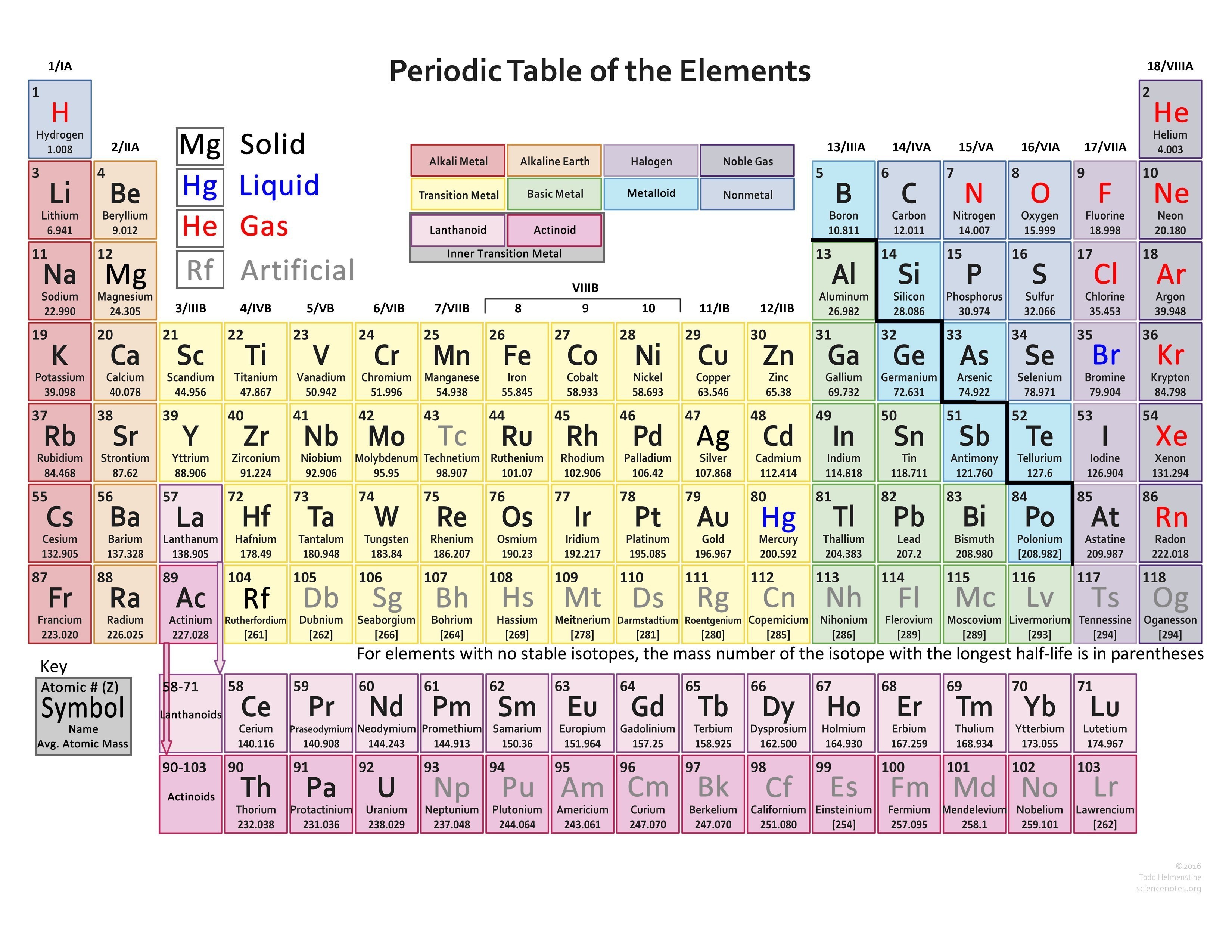
Two (or more elements) can share electrons so that each attains the electron configuration of the nearest noble gas. Nitrogen has 3 electrons int he 2p valence subshell, which is halfway This would mean it has 3 electrons of the same spin in the subshell.An element can attract additional electrons to achieve the electron configuration of the next highest noble gas.An element can give up electrons to achieve the electron configuration of the next lowest noble gas.Components with similar biochemistry are organized into groups and periods, and people from the exact same class have the identical atomic number. Achieving a Noble Gas Electron Configurationįor reasons we will discuss later, elements react until theelement achieves an electron configuration of a noble gas.Įlements can achieve a noble gas electron configuration threeways. The periodic kitchen table is a list of chemical substance aspects established in increasing buy of atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
